Lab-grown meats have received a lot of skepticism, but ready or not, lab-grown poultry is one step closer to hitting the shelves. This week, the Food and Drug Administration announced that it had completed their its consultation for UPSIDE Food’s animal cell cultured chicken — the first-ever approved lab grow meat.
Not so fast, though. The passing of this rigorous evaluation means that while UPSIDE Foods received a pass on the pre-market consultation, and we know it’s safe for human consumption, it’s not ready to hit the shelves yet.
“Since our earliest days, our top priority has been to ensure the safety and quality of our products,” said Eric Schulze, vice president of regulatory and public policy at UPSIDE Foods, according to numerous news sources. “FDA sets the standard for global acceptance of new food innovations, and we are incredibly grateful for the agency’s rigorous and thoughtful process to ensure the safety of our food supply. We’re also extremely proud to have played a leading role in helping to champion the framework for how cultivated meat, poultry and seafood are regulated in the U.S.”
In the U.S., cultivated meat is regulated by both the FDA and the U.S. Department of Agriculture. The next step in the approval process will be to secure further approvals from the USDA. Additionally, the food itself requires a mark of inspection from USDA-FSIS before it can enter the U.S. market.

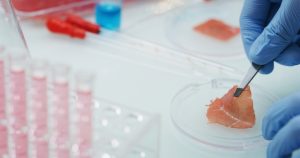
UPSIDE Foods claims that they can raise meat in a more sustainable manner than traditional agriculture, using 77 percent less water and 62 percent less land than conventional meat. The company’s website reads, “What makes our meat unique is how it’s cultivated: we take a small sample of healthy chicken cells. We place it in a nutrient-rich environment and allow it to grow into pure clean meat, ready to cook and enjoy.”
The FDA’s pre-market consultation with the firm included an evaluation of the firm’s production process and the cultured cell material made by the production process, including the establishment of cell lines and cell banks, manufacturing controls, and all components and inputs.
Here’s what the FDA has sent out so far:
- The FDA’s response letter to the firm
- The FDA’s scientific memo that describes our evaluation of the data and information provided by the firm
- The firm’s own assessment that their food is safe
The FDA says it is ready to work with additional firms developing cultured animal cell food and production processes to ensure their food is safe and lawful under the Federal Food, Drug, and Cosmetic Act. It is already engaged in discussions with multiple firms about various types of food made from cultured animal cells, including food made from seafood cells that will be overseen solely by the FDA. Human food made with cultured animal cells must meet the same stringent requirements, including safety requirements, as all other food.


